|
12/24/2023 0 Comments Carbonite formula  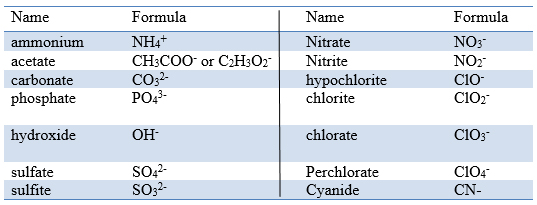
A) 28.12% B) 9.372% C) 42.73% D) 21.38% E) 35.97% 13) Write a balanced equation to show the reaction of aqueous aluminum acetate with aqueous ammonium phosphate to form solid aluminum phosphate and aqueous ammonium acetate. A) 9.83 x 1020 C2H4 molecules B) 7.74 x 1026 C2H4 molecules C) 2.71 x 1020 C2H4 molecules D) 3.69 x 1023 C2H4 molecules E) 4.69 x 1023 C2H4 molecules 12) Calculate the mass percent composition of sulfur in Al2(SO4)3. A) calcium B) cesium C) lead D) carbon E) radon 9) How many millimoles of Ca(NO3)2 contain 4.78 x 1022 formula units of Ca(NO3)2? The molar mass of Ca(NO3)2 is 164.10 g/mol. 8) Calculate the molar mass of Ca3(PO4)2. 
E) Acid rain comes from hydroelectric power plants. C) Acid rain comes from coal-powered electric power plants. A) Acid rain contains nitric acid and sulfuric acid. A) sulfuric acid B) persulfurous acid C) sulfurous acid D) hyposulfurous acid E) persulfuric acid 7) Identify the FALSE statement about acid rain. A) nitric acid B) nitrous acid C) hydrogen nitrate D) hydrogen nitriteĦ) Give the name for H2S04. A) cobalt chloride hydrate B) cobalt(1) chloride heptahydrate C) cobalt(II) chloride heptahydrate (D) cobalt(II) chloride hexahydrate E) cobalt(I) chloride 4) Give the correct formula for aluminum sulfate. A litanium(II) carbonate B) titanium carbide C) titanium carbonite D) titanium(II) carbonite E) titanium(I) carbonate 2) Which of the following is the correct chemical formula for a molecule of chlorine? A) CI B) Cl- XC) CI+ D) C12 3) Give the name for CoCl2'6H20. Remember that titanium forms several ions. Circle the correct answer 1) Give the name for TiCO3. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
